One of my local EndoWarriors has started a new medication to help with her Endometriosis pain and symptoms, based upon the recommendation of her new physician. It’s Letrozole, which I had never heard of. And she asked me if I had any info on it…so…now I’m inspired to do some research!
What is Letrozole?
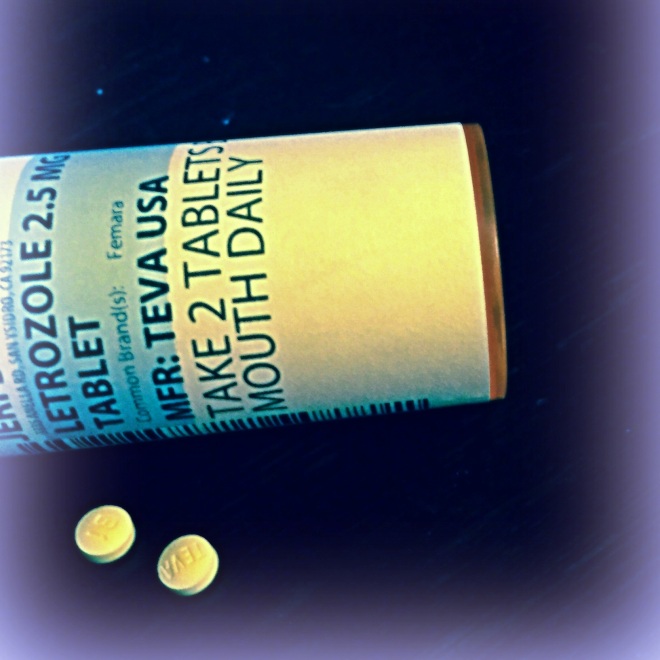
Letrozole is the generic name for Femara, a drug classified as an aromatase inhibitor. What is aromatase? It’s an enzyme that is crucial in the creation of estrogen. Aromatase inhibitors have been FDA approved for treating breast cancer in post-menopausal patients. However, it has piqued the interest of the medical community in controlling Endometriosis symptoms. It is not yet FDA approved for this treatment, but is used as an off-label, accepted treatment among the medical community. As of today’s research, there are no indications that drug manufacturers are going to seek FDA approval for aromatase inhibitors to treat Endometriosis.

Endometriosis has been theorized to be a largely estrogen-driven disease. Aromatase has been found in increased amounts in the endometrial tissue of people with Endometriosis, as opposed to people without. Some physicians are now prescribing aromatase inhibitors when no other traditional treatments have helped with Endometriosis symptoms. The hope is that by inhibiting (blocking) aromatase production, it will ease Endometriosis symptoms and progression. Aromatase inhibition may give rise to ovarian stimulation (widely used to increase fertility) and ovarian cysts; and therefore are often used in combination with other drugs, such as Norethindrone (progestin), Zoladex (GnRH agonist), or birth control pills to help patients with Endo to avoid further production of painful ovarian cysts, etc.
Side Effects
Specifically, Letrozole has been known to cause fatigue,drowsiness, and dizziness, especially when combined with alcohol or when the person is exposed to heat. A woman who is newly taking Letrozole must be cautious when driving until she truly knows how her body responds to the medication.
The FDA has released an entire list of side effects women complained of while treating with Letrozole for their breast cancer: angina (chest pain), arthritis, bone fractures, cerebrovascular/TIA (similar to a stroke), constipation, diarrhea, endometrial cancer, endometrial proliferation disorders, fractures, dizziness, edema (swelling), fatigue, headaches, hot flashes/flushes, light-headedness, myalgia (muscle pain), myocardial infarction (heart attack), nausea, night sweats, osteoporosis, other cardiovascular issues, other endometrial disorders, thromboembolic event (strokes), vaginal bleeding, vaginal irritation, vomiting, and weight increase. Some also complained of a decrease in their libido.
When used in combination with a progestin or birth control pill, aromatase inhibitors do not cause osteoporosis; however, if used with a GnRH agonist, osteoporosis may develop. Your physician may recommend frequent bone scans to ensure bone loss is not occurring while treating. They may also recommend you take Calcium and Vitamin D during your treatments.
RXList.com has an ongoing list of testimonials and reviews of patients who take Femara (Letrozole) and what their experiences and side effects have been. I know that my EndoWarriors going through a lot of joint pain at the moment. Almost everyone complained of bone and joint pain; stating:
“Then–suddenly I was having bone pain in legs (felt like someone took a 2×4 and whacked me across the leg bones)–joint pain in shoulders, knees, ankles, feet, and fingers.”
“I started having joint pain in the knees, feet, and fingers. Also fatigue and muscle weakness. Feel like I have aged 10 years.”
“I have experienced severe bone and muscle pain to the point that I find it difficult to walk up and down stairs.”
“I hurt so badly I could barely get out of bed or walk down the halls at work”
Many patients stopped taking the drug because of the intensity of the side effects and posted their reviews on RxList.
Clinical Trials
In 2004, a pilot study was published in the Fertility and Sterility journal on the effectiveness of managing Endometriosis symptoms using Letrozole. The study involved 10 women who did not respond to surgical or medical treatment of their Endometriosis. They were given 2.5mg of Letrozole, 2.5mg of Norethindrone, as well as Calicum Citrate and Vitamin D supplements for six months. The study concluded that there was a “marked reduction of laparoscopically visible and histologically confirmed endometriosis in all 10 patients and significant pain relief in nine out of 10 patients who had not responded previously to currently available treatments,” and they strongly urge the use of this regimen in treating Endometriosis symptoms.
Another study in 2004, again published in the Fertility and Sterility journal, followed the treatment of two women. They were given Anastrozole (a different aromatase inhibitor), as well as a progesterone pill, calcitriol, and rofecoxib for three months. Both of the women had a rapid reduction in their pain and symptoms, and remained so even 24 months after receiving the treatment. One underwent a diagnostic laparoscopy 15 months after treatment and was confirmed free and clear of any Endometriosis implants. Both women were able to conceive within 24 months of treatment.
The Middle East Fertility Society Journal published an article in 2014 which reviewed several different types of drugs emerging in Endometriosis treatment, including aromatase inhibitors. It also summarizes several past studies regarding Endometriosis and aromatase inhibitors.
In 2015, a study was published in BioMed Research International of a clinical trial of Letrozole and its effectiveness in decreasing the size of endometrioma (blood-filled cysts). The lead author of this study is also the doctor whom prescribed Letrozole to my EndoWarrior who inspired today’s blog. It was a three-month study of eight women, each of whom agreed to the “off-label and unproven therapy,” in the hopes it would reduce their Endometriomas and Endometriosis pain. They were given daily 5mg doses of Letrozole, as well as daily 5mg doses of Norethindrone (the FDA-approved “add back” therapy used with Lupron Depot). A typical daily dose of Letrozole is 2.5mg, but since this study was only three months long, they upped the dosage. And Norethindrone was used “…to minimize the negative effects of induced hypoestrogenemia and thus make the letrozole therapy more tolerable.” Hypoestrogenemia is a deficiency of estrogen in the blood. Over the three-month period, they found the Endometriomas decreased by 50%, and the women had significant reductions of pelvic pain and painful sex. More studies are needed, including double-blind tests with placebo, as well as longer studies, and smaller doses. It is their hope that these studies will lead to a lesser need for surgical intervention in suppressing the pain and manifestations of Endometriosis.
I encourage you to look online for a plethora of additional studies regarding aromatase inhibitors and Endometriosis.
Conclusion
Although the treatment of Endometriosis with aromatase inhibitors is not approved by the FDA, and there’s no indication that such approval will be sought, the studies and trials do appear to support the notion that they help reduce and control the pain and progression of this disease. If you are able to swallow the idea of an experimental drug, you may want to talk to your physician about this treatment regimen.
(Updated March 27, 2019)
Resources:
Advanced Fertility Center of Chicago
Fertility and Sterility (2004; Article) : Treatment of Endometriosis and Chronic Pelvic Pain with Letrozole and Norethindrone Acetate : A Pilot Study
Fertility and Sterility (2004; Article) : Successful Treatment of Severe Endometriosis in Two Premenopausal Women with an Aromatase Inhibitor
Biomed Research International (2015; Article) : Reduction of Endometrioma Size with Three Months of Aromatase Inhibition and Progestin Add-Back
Northwestern University Feinberg School of Medicine
Middle East Fertility Society Journal (2014: Article) : Emerging Treatment of Endometriosis
U.S. Food and Drug Administration : Femara (Letrozole tablets) drug information page
~Again, I am a layman. I do not hold any college degrees, nor mastery of knowledge. Please take what I say with a grain of salt. If curious, do your own research 😉 Validate my writings. Or challenge them. And ALWAYS feel free to consult with your physician (or in this case, attorneys). Always. Yours ~ Lisa



I can’t thank you enough for posting this Lisa!
This being my third day on this medication, I can’t provide too much. But the side effects that I have had over the last few days are quite unpleasant.
Endo symptoms, as well as tests:
Pelvic and thigh radiating, aching pain
Extreme fatigue
Cramping
Sharp pains on lower right side
Dull sharp pain on left side
Rectal pain
Sciatic nerve pain
Increased tendinitis pain in my hips
Pain with intercourse
Light bleeding over the course of months
TESTING:
Transvaginal Ultrasound showing small endometrioma on right ovary, and a very visible endometriosis strip
Pelvic exam extremly painful but did give my doctor the idea that I may possibly have endometriosis on my rectum, so I am being referred to a GI.
With that being said I am on Letrozole 5mg daily and Depo.
While the medication has decreased my cramping and I have yet to have bleeding; the side effects are becoming debilitating.
Extreme joint pain everywhere (even my toes)
Very tired about an hour after taking medication
Slight headache
Nervous/anxious feeling
Loss of appetite.
Again, I just started this medication so there is still hope that my body will get acclimated to it.
Keep your head up EndoSisters, we will get through this. (Even if I have to use a walker lol)
LikeLiked by 1 person
I have been on this medication for a little over a month and have noticed a drastic change in my endo pain. My story with endometriosis starts, who knows, probably since I was born but I didn’t start having the knife stabbing/twisting pain that occurred outside of my ‘time of the month’ until around 2007. Had my first lap in 2008 (one of the lucky ones that found out early). I already had my one daughter, who was around 3, when I first having ‘the pains’. I was determined to have at least one more child before I ruined any of my chances or so I thought might. So I finally met the love of my life in 2009 so didn’t waste any time and after trying for only 5 months we had my 2nd daughter in 2010. The endo ‘behaved’ for almost 4 years after the birth of my 2nd daughter but it came back with vengeance!! Instead of creeping on like the first time it seemed to come on with greater intensity and persistency. Mentioned the return of pain to my GYNO at my yearly visit and she orders an Ultrasound which shows a grapefruit size cyst on one ovary and a lemon sized cyst on the other. She gave me the option to either keep an eye on them or have a laporoscopy. At that point I was having so much pain I decided on the laporoscopy to see what was going on in there per say. So had the lap done in november 2014 where my GYNO found “endo everywhere” but felt she wouldn’t even scratch the surface if she tried to remove any of it. So our next visit was the discussion to have a hysterectomy “because that will cure my endometriosis” as she would think. So I had a hysterectomy LAVH with BSO (Lap Assist Vaginal Hysterectomy with Bilateral Salpingoophrectomy) on December 31st, 2014. The recovery was rough, didn’t think I’d ever feel ‘normal’ again. Around the 7-8 week mark (when my OBGYN thought I should be returning to work) I started noticing those oh so familiar pains again. I returned to work 12 weeks after my hysterectomy with those same pains that I was counting down the days of having before I left for the surgery! So I decided to see a specialist Dr. Patrick Yeung from SLU hospital in St. Louis and with some trial and error from myself with diet (found avoiding caffiene, fried foods, gluten, and dairy helped) and tried norethindrone for 3 weeks, which only made me feel like I had PMS with the irritability and exhaustion and still that pain every 3 weeks(where it didn’t matter what I ate or drank) as if I was having a period. So he started me on this Femara around July 14th, 2015. I take it at night before I go to bed. I don’t notice the dizziness as a side effect but it didn’t take long (maybe a week or 2) to notice the hot flashes and night sweats. I have had back/body aches for as long as I can remember but I do notice more intense pain in my back and shoulders. I also notice joint pain in my fingers. But the ‘endo pain’ is soo much improved. I find it takes alot more of those food/drink triggers to cause the pain so I can eat/drink those things in moderation right now. I’ve had 2 of those ‘every 3 week’ period like cycles and instead of 5-10 days of misery it has been more like 1-2 days of sporatic pain, not the all day pain like before. All in all my qualitiy of life as soo much improved!! I have always been too stubborn to call into work (as an RN on a busy outpatient surgery unit) and even though with the expense of the diet, meds I take, my kids, and the rising costs of everything I need to work the 40 hours a week, I can push myself through the 30(3- 10 hour shifts) per week. I hope this helps others as much as it’s helped me!
LikeLiked by 1 person
So a month later I’ve decided to stop taking the Femara, so I was on it for a total of 2 months. Like I said I noticed much improvement in the ‘endo’ pain with in 2 weeks but the back pain and memory problems were getting worse and worse as I was on the med! I couldn’t even walk without the pain and motrin was the only thing that made it at least bearable for me! I also felt as if I was coming down with Alzheimers where I couldn’t even remember extension numbers to departments at the hospital I’ve worked at for the last 9 years. So I stopped taking the Femara last monday was my last dose and here it is the thursday after (yes a week and a half later) that I finally have gone 24 hours without any pain meds and its bearable to walk. So far the endo pain is still behaving (compared to what it used to be) but I will keep you updated. I was really craving a soda which always puts me in pain and after drinking a 32 oz fountain soda the pain started but as I am sitting and typing this the pain is fading where before that soda would’ve haunted me before the femara!! So crossing my fingers it stays this way!!
LikeLiked by 1 person
Oh man, I’m sorry to hear it caused such issues, but am very glad you honored your body and stopped taking it. Hoping for a good outcome for ya…
LikeLike
Thank you all for contributing. It’s really hard to find out info about this treatment from women with endo.
I just started Anastrozole + necon birth control last month for my endo. I was urged to try it out by Dr. Bulon at Northwestern rather than having a hysterectomy.
I don’t think it’s been long enough to give a full review of the drug combination (it apparently takes 6months to really notice improvements) but as of now I’m extremely tired, I feel like my body temp has risen – ESP when active or at night sleeping. Also, I’ve noticed that my muscles are randomly twitching which has never happened before. Lastly, I’m experiencing a new form of cramping. My pelvis feels sore, tight and constantly radiating a dull pain. Usually, I experience sharp stabbing and twisting feelings.
I’m willing to give this a try b it’s my last available option but as of now I can’t say that it has improved or changed my quality of life – & all I want to do is nap! I can barely keep my eyes open.
Thanks again for writin this article! The more we share, the more we know, the faster we can find a cure! X
LikeLike
❤ You are so welcome! Thank you for your feedback, and your personal experiences!! I hope as the treatment continues the side effects lessen and you find a greater quality of life. Keep us posted!
LikeLike
I do not have a surgical diagnosis of endometriosis, but have had blood filled cysts on my ovaries for years and very painful periods. Last few included severe leg pain, and at the same time leg numbness (which made me super freak out). My gynecologist started me on 30 mg of provera every day, which made me completely insane and even suicidal. I dropped to 10 mg, but then I got my period. New doctor prescribed 2.5 mg per day of letrozole and 2.5 mg of norethindrone twice a day. I’ve only been on it for 2 weeks, but I feel AWESOME! I am worried though that it wont be enough to keep my period away. Anybody know if it will?
LikeLiked by 1 person
Thank you so much for sharing this! My doc just talked to me yesterday about this drug. I’m skeptical about trying it. I have had 10 surgeries in the last 5 years.
Never had children and I want a child. So she wants me to use this to help my fertility but also keeping the endo at bay. But these side effects….it’s like a complete lose/lose situation. Guess I will give it try! Stay strong ladies!! #endowarriors
LikeLike
Hi Tinker. Just following up with you. Did you decide to go forward with the treatment? If so, how are you feeling? And if you didn’t go forward with the treatment, how are you feeling? 😉 I hope well. Yours, Lisa
LikeLike
Hi! Thanks for asking. I made the choice not to take the meds. Lots of things are going on now with lots of pain. I ended up seeing a specialist a couple weeks ago and found out I’m eat up with endo again. (I have stage 4) Actually right now I’m having new symptoms with pain on my sciatic nerve and hurting while breathing. Got to wait until consultation to share that. Why can’t someone find a cure?
LikeLike
I am SO sorry to hear this!!! But I’m right there with ya; waiting on a consult with my physician on the 16th to discuss my new pain, too. Meh. I’m glad you didn’t go through with the meds; they sound miserable. One day someone will find a cure. Maybe just not in our lifetime. 😦 Hang in there, hun. Best of luck to ya for your upcoming appointment!
LikeLike
I have tried so many medicines over the years that were supposed to be so great for endo, I just couldn’t stand to try one more that had so many side effects. Thanks for the kind words and support! It’s nice to hear words of support from others who are going through the same thing. I wish you the best at your next appointment! Stay strong!! #endowarriors
LikeLiked by 1 person
I used Femara years ago, when I still had my ovaries. I had a complete hysterectomy at age 26. I am now 34 and still have Stage 4 Endo. I had 4.5 years pain free after my hysterectomy. I didn’t have any side-effects worse than my Endo pain, I can’t actually remember if I had any at all. I’ve had 5 surgeries and tried every pill, shot… whatever under the sun. Femara did stop my pain; for 3 months. Then my body absorbed it and went back to functioning as ‘normal’ (same goes for provera & lupron shots). This drug is for post-menopausal women (I was warned it may not play well with the ovaries), so I would actually be up to try it again but it’s SO expensive and insurance won’t cover off-label use. I WISH I had used Femara right away after my hysterectomy to starve out any left over disease – I probably would be much better off than I am today.
LikeLike
Stacey, I am so sorry to hear about your ongoing pain. I hope you can find some type of relief.
LikeLike
I took this for a month to ‘try’ it instead of my BC pill. My body HATES and HATED not being on the BCP – even if I forget to take it at the same time my body goes into pain mode to remind me. I can’t remember much about being on the letrozole (but I wrote some notes somewhere) but I know I was in a lot of pain at first. Then as soon as I stopped it I got a blood-filled cyst! Never had a cyst before, think it was an endo-related cyst and blame these drugs. Also drugs where you need to take Calcium and Vitamin C not to damage your bones? No thank you!
LikeLiked by 1 person
A month typically isn’t enough time for it to take full effect for pain. It’s like switching birth control pills, they tell you to stay on it for 6 months minimum tonsee the difference. No birth control ever worked, lupron and depo provera have the same side effects with the bone density. The Femara works at a different level. It stops even the estrogen that lives in the Endo modules. When the pain is unbearable, for me the side effects are worth it.
LikeLiked by 1 person
Totally agree a month is not long enough but for whatever reason the docs did not pursue it with me. As soon as I stop taking any of these things my pain is right back. Aren’t these things supposed to suppress the estrogen for longer?
LikeLike
Any anti estrogen pills, like Femara won’t stay in the system long when you stop taking them. It’s been proven that women with Endo have more estrogen than women without. Its not just produced in our ovaries, doctors keep trying to suppress only that. So when even the Endo implants realize they aren’t suppressed anymore, production begins right away and sometimes more (because even though on these suppressing drugs, Endo can still grow in other parts of the body. It breaks off and goes through the blood stream). Then our pain is worse!
LikeLike